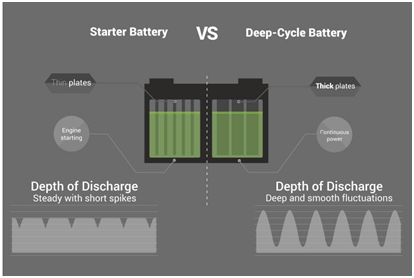
In the dynamic world of automotive engineering, every component plays a crucial role in determining the overall performance of a vehicle. Among these components, the automotive battery stands out as a silent powerhouse, providing the electrical energy necessary to start the engine and power various onboard systems. At Sarex, a reliable automotive battery manufacturer, we understand the pivotal connection between our batteries and vehicle performance, driving innovation to ensure optimal functionality and reliability.
Reliability underpins the core ethos of Sarex automotive batteries. A vehicle’s ability to start consistently and reliably is paramount for its performance, especially during critical moments. Whether it’s a frigid morning in winter or a scorching afternoon in summer, drivers rely on their batteries to kickstart their engines without fail. With cutting-edge technology and stringent quality control measures, Sarex batteries are engineered to deliver unrivaled reliability, ensuring that vehicles start swiftly and operate smoothly in all conditions.
Moreover, the performance of modern vehicles is increasingly reliant on electrical systems for functions ranging from infotainment and navigation to advanced driver-assistance systems (ADAS). As vehicles become more technologically advanced, the demand for reliable power sources escalates. Sarex automotive batteries rise to the challenge, providing a stable and consistent power supply to support these intricate electrical systems. By maintaining optimal voltage levels, Sarex batteries enable seamless operation of onboard electronics, contributing to enhanced comfort, safety, and convenience for drivers and passengers alike.
Efficiency is another key aspect where Sarex automotive batteries shine. An efficiently functioning battery ensures that the vehicle’s electrical system operates at peak performance, maximizing fuel efficiency and reducing emissions. By minimizing energy wastage and optimizing power delivery, Sarex batteries contribute to a more eco-friendly driving experience, aligning with the growing emphasis on sustainability within the automotive industry.
In the realm of performance vehicles, where power and precision are paramount, Sarex automotive batteries continue to make their mark. Whether it’s a high-performance sports car or a rugged off-road vehicle, the demands placed on the battery are heightened. Sarex rises to the challenge by engineering batteries capable of delivering the high cranking power and enduring performance required by such vehicles. With advanced construction techniques and premium materials, Sarex batteries provide the robustness and resilience needed to withstand the rigors of intense driving conditions, ensuring that performance enthusiasts can push their vehicles to the limit with confidence.
Furthermore, the reliability and durability of Sarex automotive batteries extend their utility beyond vehicles. In the rapidly expanding market for home and e-rickshaws, where battery technology reigns supreme, Sarex is at the forefront of innovation. By leveraging expertise in energy storage and electrochemistry, Sarex develops cutting-edge batteries tailored to the specific requirements of e-rickshaws. These batteries offer superior energy density, fast charging capabilities, and an extended lifespan, enhancing the overall performance and viability of electric mobility solutions and powering homes for a longer time.
In conclusion, the connection between Sarex automotive batteries and vehicle performance is indisputable. Through relentless innovation, uncompromising quality, and unwavering reliability, Sarex continues to set the benchmark for excellence in battery technology. As vehicles evolve and embrace new technologies, Sarex remains steadfast in its commitment to powering the future of automotive performance.